What's the take-home message from this long-term data?īrahmer: With 5 years of follow-up, pembrolizumab continues to show meaningful improvements in OS and durable response compared with chemotherapy. 
There was no change since the updated analysis at 25 months. Seven additional patients in the pembrolizumab arm, and no additional patients in the chemotherapy arm, had grade 3-5 AEs since the initial publication of KEYNOTE-024. What about long-term adverse events (AEs)?īrahmer: Fewer patients in the pembrolizumab group experienced treatment-related AEs. The updated analysis of median OS showed a 38% reduction in the survival hazard with pembrolizumab (95% CI 0.48-0.81). In earlier data, the pembrolizumab arm had a median progression-free survival (PFS) of 10.3 months versus 6.0 months for the chemotherapy arm ( P<0.001). The results also are consistent with the KEYNOTE-001 trial, a nonrandomized trial that showed a 5-year OS rate of 29.6% with single-agent pembrolizumab in patients with treated and untreated metastatic NSCLC. The results confirmed and strengthened observations from a previously reported analysis of 3-year survival. 
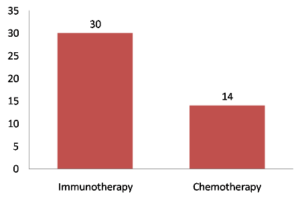
That advantage with the PD1 inhibitor held fast even though two-thirds of patients in the chemotherapy group ultimately crossed over to the immunotherapy arm, which might have led to underestimation of the magnitude of benefit with pembrolizumab, the authors noted. Specifically, 5-year OS came in at 31.9% in pembrolizumab-treated patients and 16.3% in chemotherapy-treated patients so that the median OS was twice as long with pembrolizumab at 26.3 months versus 13.4 months. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
